Do Noble Gases Form Ions
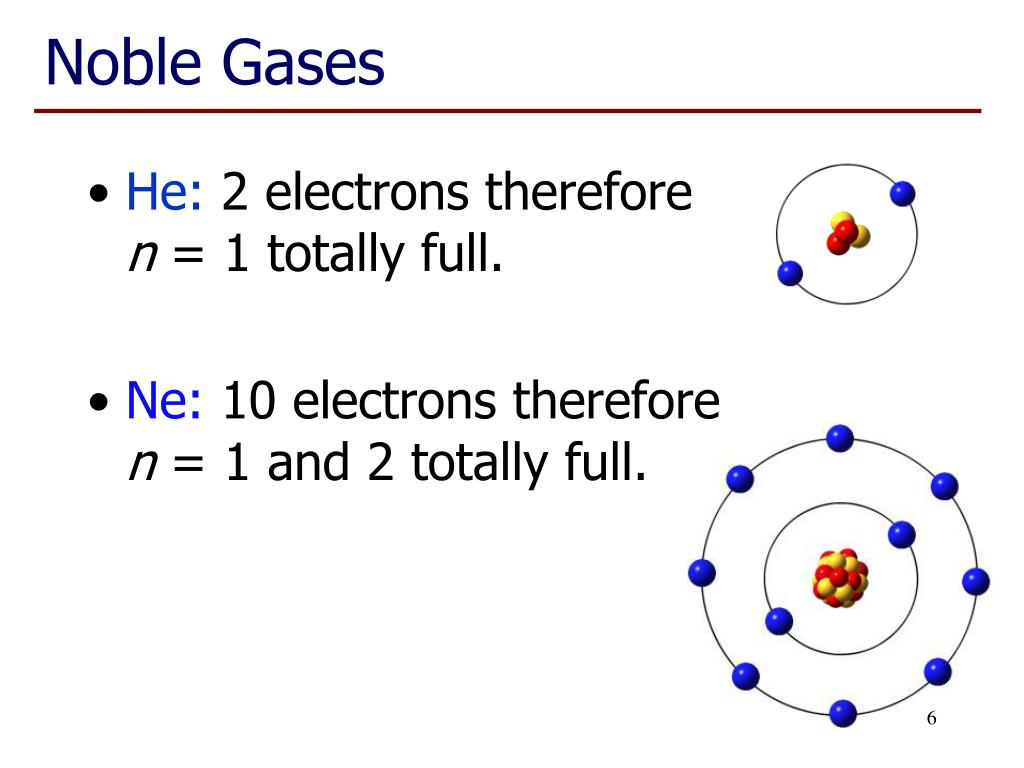
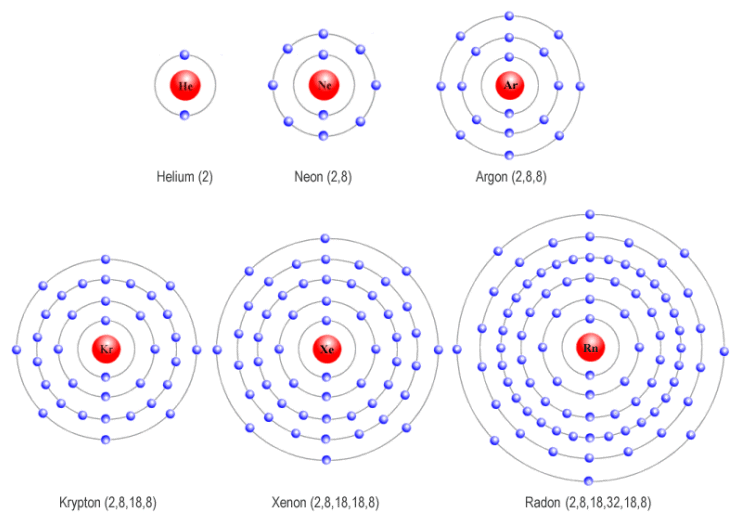
Do Noble Gases Form Ions - This trend can be used as a guide in. Electron configurations shells, subshells, and orbitals introduction to electron configurations noble gas. The elements are helium (he), neon (ne), argon (ar), krypton. Web moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble. This is why the noble gases are inert and do not take part. Web noble gases (group 8): Web here are five of the six noble gases: The full valence electron shells of these atoms make. Web the atoms of noble gases already have complete outer shells, so they have no tendency to lose, gain, or share electrons. Web ionic bonding is the complete transfer of valence electron (s) between atoms and is a type of chemical bond that generates two oppositely charged ions.
Web moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble. Web argon chemistry atomic argon has a filled shell configuration with filled 3s and 3p orbitals. Web here are five of the six noble gases: Helium, neon, argon, kypton and xeon. Group 16 elements (two groups left) form 2− ions, and so on. They're all colourless and transparent. The elements are helium (he), neon (ne), argon (ar), krypton. There are special names for certain types of ions. For neutral atoms, the 3d orbitals are above the 4s orbitals in energy. Web gcse wjec group 0 and testing ions flame tests identify alkali metal ions in compounds.
0 (uncharged) dianions, dications, and zwitterions. Web the atoms of noble gases already have complete outer shells, so they have no tendency to lose, gain, or share electrons. Web here are five of the six noble gases: Web gcse wjec group 0 and testing ions flame tests identify alkali metal ions in compounds. The noble gases of the periodic table do not have a charge because they are nonreactive. Group 16 elements (two groups left) form 2− ions, and so on. Web argon chemistry atomic argon has a filled shell configuration with filled 3s and 3p orbitals. Web they are helium, neon, argon, krypton, xenon, and radon. Web ionic bonding is the complete transfer of valence electron (s) between atoms and is a type of chemical bond that generates two oppositely charged ions. Chemistry library > unit 7 lesson 4:
Ionic Compound Nomenclature Presentation Chemistry
Krypton and xeon form compounds only with. Web noble gases (group 8): The noble gases are very unreactive. Web moving from the far right to the left on the periodic table, elements often form anions with a negative charge equal to the number of groups moved left from the noble. Web argon chemistry atomic argon has a filled shell configuration.
Ionic Compound Nomenclature Presentation Chemistry
The elements are helium (he), neon (ne), argon (ar), krypton. Helium, neon, argon, kypton and xeon. Electron configurations shells, subshells, and orbitals introduction to electron configurations noble gas. The noble gases are very unreactive. Krypton and xeon form compounds only with.
What Are the Freezing, Melting, and Boiling Points of Solids, Liquids
0 (uncharged) dianions, dications, and zwitterions. Web they are helium, neon, argon, krypton, xenon, and radon. For neutral atoms, the 3d orbitals are above the 4s orbitals in energy. Krypton and xeon form compounds only with. Web noble gas, any of the seven chemical elements that make up group 18 (viiia) of the periodic table.
Why do Noble Gases rarely form Bonds with other Atoms? MakeTheBrainHappy
Helium, neon, argon, kypton and xeon. There are special names for certain types of ions. This trend can be used as a guide in. Web noble gases (group 8): Web gcse wjec group 0 and testing ions flame tests identify alkali metal ions in compounds.
10 28 How Many Electrons Do Atoms Gain Lose
This trend can be used as a guide in. Part of chemistry (single science) atomic. Web noble gas, any of the seven chemical elements that make up group 18 (viiia) of the periodic table. Chemistry library > unit 7 lesson 4: Krypton and xeon form compounds only with.
PPT Recap Atomic Structure PowerPoint Presentation, free download
0 (uncharged) dianions, dications, and zwitterions. Web they are helium, neon, argon, krypton, xenon, and radon. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. For neutral atoms, the 3d orbitals are above the 4s orbitals in energy. Web gcse wjec group 0 and testing ions flame tests identify alkali metal ions in compounds.
Trends on the Periodic Table Chemistry Is My Jam!
0 (uncharged) dianions, dications, and zwitterions. Electron configurations shells, subshells, and orbitals introduction to electron configurations noble gas. Web what charge do noble gases take as ions? Group 16 elements (two groups left) form 2− ions, and so on. For neutral atoms, the 3d orbitals are above the 4s orbitals in energy.
Chemistry 9/15, 9/18 Noble Gas Configurations, The Atomic Museum
Web the ions have the electronic structure of a noble gas (group 0 element), with a full outer shell for elements in groups 6 and 7, the charge on the ion is equal to (8 minus group. This is why the noble gases are inert and do not take part. Part of chemistry (single science) atomic. Web under certain conditions,.
Chemical Bonding
Web noble gas, any of the seven chemical elements that make up group 18 (viiia) of the periodic table. Web for example, group 17 elements (one group left of the noble gases) form 1− ions; Web argon chemistry atomic argon has a filled shell configuration with filled 3s and 3p orbitals. Web what charge do noble gases take as ions?.
Web Noble Gases Are Odorless, Colorless, Nonflammable, And Monotonic Gases That Have Low Chemical Reactivity.
For neutral atoms, the 3d orbitals are above the 4s orbitals in energy. Web noble gas, any of the seven chemical elements that make up group 18 (viiia) of the periodic table. 0 (uncharged) dianions, dications, and zwitterions. Group 16 elements (two groups left) form 2− ions, and so on.
Electron Configurations Shells, Subshells, And Orbitals Introduction To Electron Configurations Noble Gas.
Web ionic bonding is the complete transfer of valence electron (s) between atoms and is a type of chemical bond that generates two oppositely charged ions. Part of chemistry (single science) atomic. Krypton and xeon form compounds only with. Web noble gases (group 8):
A Noble Gas Configuration Of An Atom Consists Of The Elemental Symbol Of The Last Noble Gas Prior To That Atom, Followed.
There are special names for certain types of ions. They're all colourless and transparent. The elements are helium (he), neon (ne), argon (ar), krypton. This is why the noble gases are inert and do not take part.
Web The Atoms Of Noble Gases Already Have Complete Outer Shells, So They Have No Tendency To Lose, Gain, Or Share Electrons.
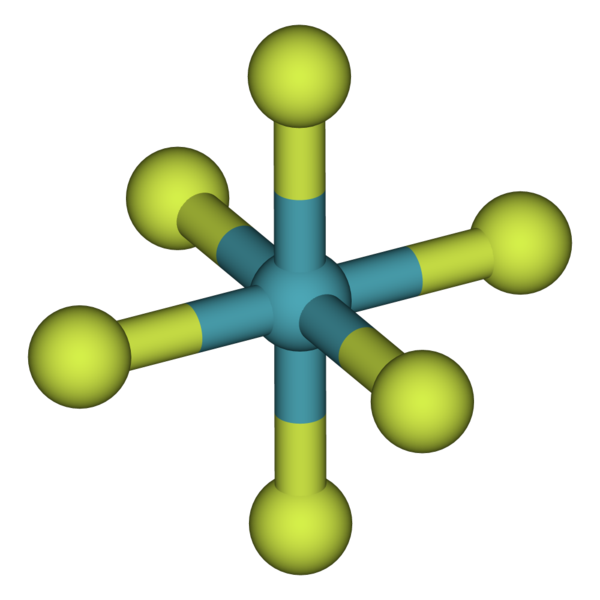
This trend can be used as a guide in. Web the ions have the electronic structure of a noble gas (group 0 element), with a full outer shell for elements in groups 6 and 7, the charge on the ion is equal to (8 minus group. Web under certain conditions, the noble gases can form diatomic gases, clathrates, fluorides, chlorides, metal complexes, and other compounds. Web for example, group 17 elements (one group left of the noble gases) form 1− ions;
.PNG)
.PNG)