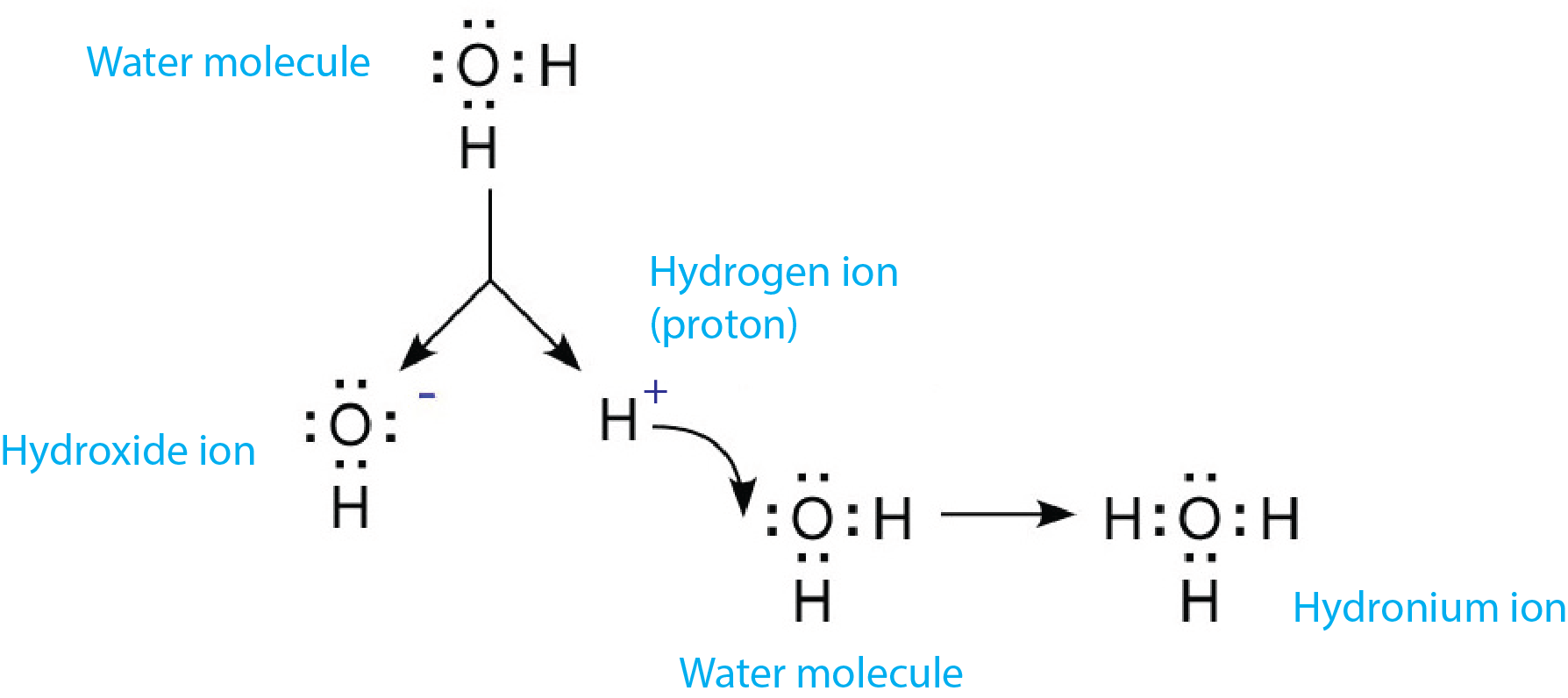
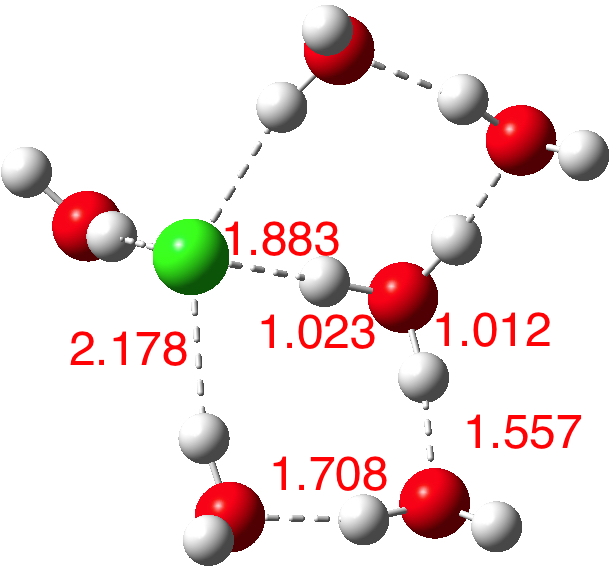
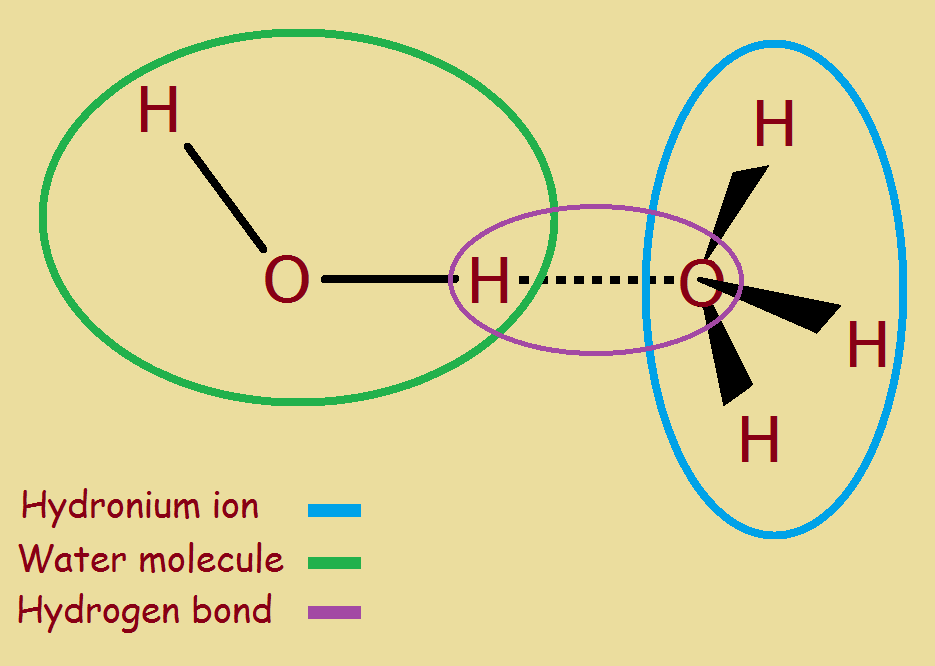
How Does Hydronium Ion Form
How Does Hydronium Ion Form - Web in aqueous solution of acids, acid dissociate to form hydrogen ions and combines with water molecules to form hydronium ion (h3o+). The concentration of h₃o⁺ in a strong acid solution is. Learn about the hydronium cation, which has the chemical formula h3o+. Web hydronium ion definition, the hydrogen ion bonded to a molecule of water, h3o+, the form in which hydrogen ions are found in aqueous solution. Solution when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron. It’s chemical formula is h3o+. Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be. Web it can be formed when an acid is present in water or simply in pure water. Chapter 5 / lesson 24. Web [1] in aqueous media, the free acid cation is mostly present as the hydrated proton (h 3 o) + but for convenience it is often referred to as a hydrogen ion or proton (h + ), and thus the.
Solution when a protic acid is added to water, it releases a hydrogen ion (h+) (also called aproton because a hydrogen atom missing one electron. It can also be formed by the combination of a h+ ion with an h2o. Web $\begingroup$ about the charge: Web [1] in aqueous media, the free acid cation is mostly present as the hydrated proton (h 3 o) + but for convenience it is often referred to as a hydrogen ion or proton (h + ), and thus the. Calculate the hydronium ion concentration of human blood. Web it can be formed when an acid is present in water or simply in pure water. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Web strong acids (such as hcl, hbr, hi, hno₃, hclo₄, and h₂so₄) ionize completely in water to produce hydronium ions. Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be. Web the hydronium ion (h 3 o +), which is the hydrogen ion in aqueous solution, also belongs to this class.
Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be. Web the hydronium ion (h 3 o +), which is the hydrogen ion in aqueous solution, also belongs to this class. Web hydronium is what you get when you put water and hydrogen ions together, forming h 3 o +. It’s chemical formula is h3o+. Hydronium is the key to calculating. Web strong acids (such as hcl, hbr, hi, hno₃, hclo₄, and h₂so₄) ionize completely in water to produce hydronium ions. Calculate the hydronium ion concentration of human blood. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Web $\begingroup$ about the charge:
Hydronium Alchetron, The Free Social Encyclopedia
Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. The charge of these ionic acids, of course, always must be balanced by. Web $\begingroup$ about the charge: Web the hydronium ion (h 3 o +), which is the hydrogen ion in aqueous solution, also belongs.
Hydronium Ion Formation and Lewis Dot Structure YouTube
Web the hydronium ion (h 3 o +), which is the hydrogen ion in aqueous solution, also belongs to this class. 0 + (+1) = +1, i.e. Web it can be formed when an acid is present in water or simply in pure water. Calculate the hydronium ion concentration of human blood. It can also be formed by the combination.
Multimedia pH and Color Change Chapter 6, Lesson 8 Middle School
0 + (+1) = +1, i.e. Hydronium is the key to calculating. The concentration of h₃o⁺ in a strong acid solution is. Web the hydronium ion (h 3 o +), which is the hydrogen ion in aqueous solution, also belongs to this class. Web it can be formed when an acid is present in water or simply in pure water.
How many water molecules does it take to ionise HCl? « Henry Rzepa
The concentration of h₃o⁺ in a strong acid solution is. Web $\begingroup$ about the charge: Web hydronium ion definition, the hydrogen ion bonded to a molecule of water, h3o+, the form in which hydrogen ions are found in aqueous solution. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}..
H3O+, l'ion hydronium définition et explications
Web [1] in aqueous media, the free acid cation is mostly present as the hydrated proton (h 3 o) + but for convenience it is often referred to as a hydrogen ion or proton (h + ), and thus the. Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields.
Molecular Facts and Structures Chemical structure, Structure
Web the thing is an acid is any substance that increases the hydronium ion concentration of the solution or yields hydronium ions on dissociation. Calculate the hydronium ion concentration of human blood. 0 + (+1) = +1, i.e. It’s chemical formula is h3o+. If not, what information do you need in order to calculate it?
Hydronium Ion Easy Science Easy science, Water molecule, Molecules
Web in aqueous solution of acids, acid dissociate to form hydrogen ions and combines with water molecules to form hydronium ion (h3o+). It can also be formed by the combination of a h+ ion with an h2o. 0 + (+1) = +1, i.e. Web it can be formed when an acid is present in water or simply in pure water..
Hydronium Ion Definition & Formula Video & Lesson Transcript
Web hydronium ion definition, the hydrogen ion bonded to a molecule of water, h3o+, the form in which hydrogen ions are found in aqueous solution. Learn about the hydronium cation, which has the chemical formula h3o+. Web hydronium is what you get when you put water and hydrogen ions together, forming h 3 o +. If not, what information do.
Hydronium wikidoc
The concentration of h₃o⁺ in a strong acid solution is. Web it can be formed when an acid is present in water or simply in pure water. Learn about the hydronium cation, which has the chemical formula h3o+. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Thus, in.
Hydronium Ion Hydrate Its Cause and Molecular Structure
Calculate the hydronium ion concentration of human blood. Web [1] in aqueous media, the free acid cation is mostly present as the hydrated proton (h 3 o) + but for convenience it is often referred to as a hydrogen ion or proton (h + ), and thus the. 0 + (+1) = +1, i.e. Web hydronium ion definition, the hydrogen.
Web The Hydronium Ion (H 3 O +), Which Is The Hydrogen Ion In Aqueous Solution, Also Belongs To This Class.
Web [1] in aqueous media, the free acid cation is mostly present as the hydrated proton (h 3 o) + but for convenience it is often referred to as a hydrogen ion or proton (h + ), and thus the. Web strong acids (such as hcl, hbr, hi, hno₃, hclo₄, and h₂so₄) ionize completely in water to produce hydronium ions. Learn about the hydronium cation, which has the chemical formula h3o+. Hydronium is the simplest form of oxonium, which is any ion that.
It’s Chemical Formula Is H3O+.
Chapter 5 / lesson 24. Calculate the hydronium ion concentration of human blood. Web hydronium is what you get when you put water and hydrogen ions together, forming h 3 o +. 0 + (+1) = +1, i.e.
Thus, In Aqueous Solution Only Can A.
Web hydronium ion definition, the hydrogen ion bonded to a molecule of water, h3o+, the form in which hydrogen ions are found in aqueous solution. Web hydrodium ions are the combination of a water molecule and a hydrogen ion, resulting in the formula, {eq}h_{3}o^{+} {/eq}. Hydronium is the key to calculating. Web how hydronium ion is formed?
The Charge Of These Ionic Acids, Of Course, Always Must Be Balanced By.
If not, what information do you need in order to calculate it? Web $\begingroup$ about the charge: Water is uncharged, the hydrogen ion is charged, so the hydronium ion as the product of the two will be. Web it can be formed when an acid is present in water or simply in pure water.